苄氧羰基(Cbz)保护氨基 |
您所在的位置:网站首页 › 苄氧羰基脱保护 › 苄氧羰基(Cbz)保护氨基 |
苄氧羰基(Cbz)保护氨基
|
除Cbz-Leu为油状物外,绝大多数氨基酸的苄氧羰基衍生物都可以得到结晶。有的N-苄氧羰基氨基酸能同它的钠盐按一定比例形成共晶,共晶产物的熔点较高,并难溶于有机溶剂。例如,苯丙氨酸经苄氧羰基化后再加酸析出Cbz-Phe时往往得到共晶产物(熔点144℃),此共晶产物用乙酸乙酯和1M HCl一道震摇时可完全转化为Cbz-Phe而溶于乙酸乙酯中。因此。除Cbz-Gly以外,一般都是采用酸化后用有机溶剂提取的方法来得到纯的N-苄氧羰基氨基酸。
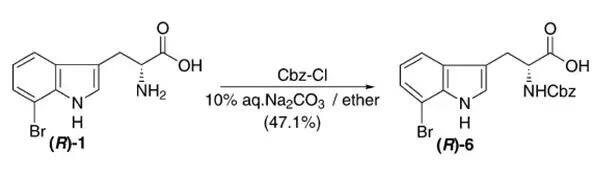
Cbz-Cl (18.5 μl,0.155 mmol) in diethyl ether (0.2 ml) was dropped to a solution of (R)-1(36.4 mg, 0.129 mmol) in 10% aqueous Na2CO3(1.8 ml) at 0°C,and stirred for 5 h. The reaction mixture was acidified with 10% citric acid,extracted with CHCl3(10mlX3). The organic layer was washed with water, driedover Na2SO4, evaporated to give light yellow gels, which werepurified by preparative TLC (CHCl3/MeOH=5:1) to afford (R)-6 (25.7 mg, 47.1%) as yellowamorphous solid. 【Konda-Yamada,Yaeko; Okada, Chiharu et al., Tetrahedrom; 2002, 58(39),7851-7865】
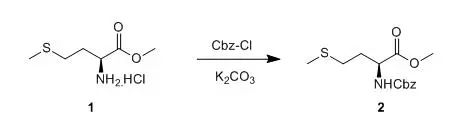
A 3-L, three-necked, Mortonflaskequipped with an efficient mechanicalstirrer, thermometer, and a dropping funnelis charged with L-methioninemethyl ester hydrochloride 1 ( 117.6 g , 0.56 mol), potassiumbicarbonate ( 282.3 g , 2.82mol, 5 eq.), water (750 mL), and ether(750 mL),and the solution is cooled to 0°C .Benzyl chloroformate( 105 g , 88.6 mL, 0.62 mol, 1.1 eq.)isadded dropwise over 1 hr, the cooling bathisremoved, and the solution is stirred for 5 hr. Glycine( 8.5 g , 0.11mol, 0.2 eq.)is added (to scavenge excess chloroformate) and the solutionis stirred for an additional 18 hr. The organic layer is separated, and theaqueous layer is extracted with ether(2 × 200 mL). The combined organic layers are washedwith 0.01 Mhydrochloric acid(2 × 500 mL), water (2 × 500 mL), and saturated brine(500 mL), and thendried (Na2SO4), filtered, and evaporated on a rotary evaporator. The resulting oil is further dried ina Kugelrohr oven( 50°C , 0.1 mm ,12 hr) to leave product 2as a clear oil that solidifies upon cooling: 165– 166 g (98–99%), mp 42– 43°C . 【M. Carrasco, R. J. Jones, S. Kamelet a1., Org.Syn.,70, 29】
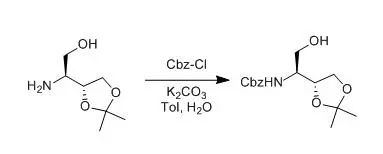
To a mixture oftoluene (3.85 L),water (3.85 L), andK2CO3(470 g, 3.40 mol) were successively added 1a(770 g, 2.72 mol) and CbzCl (488 g, 2.72 mol) with vigorous stirring at atemperature below 25 °C. After stirringat room temperature for 3 h, triethylamine (27.5 g, 270 mmol) and NaCl (578 g) were successively added, and the mixture wasstirred for a further 30 min. The organic layer was separated and concentratedto give the desired productas oil, which was used for the next reactionwithout purification. The analytical sample was prepared by columnchromatography。 【Inaba, Takashi; Yamada, Yasuki et al J. Org. Chem.,2000, 65(6), 1623-1628】 未完待续 本文非原创内容,版权归原作者所有。返回搜狐,查看更多 |
【本文地址】
今日新闻 |
点击排行 |
|
推荐新闻 |
图片新闻 |
|
专题文章 |